- Home
- Weddings
- Portraits
- Journal
- Contact
- Single line font in fusion 360
- Cultist simulator wiki moth
- Hollywood movie hindi dubbed hd 720p download
- A n eutron changes into a proton and an electron
- Adblink download windows
- Enable gmail desktop notifications
- The hidden face movie
- Abirami anthathi in tamil pdf
- Clipp store 2019 suporte
- The wailers - bob marley ganja gun
- Ghost32-7z
- Alien skin exposure 7 taringa
- Passware kit professional 2015 v-3
- Easeus data recovery torrent
- Best unknown nes games
- Dxcpl directx 11 emulator windows 10
- Cell card games
- Sims 3 midnight hollow tumblr
- Corel paintshop pro x6
- Hyper dragon ball z gohan move list
- Windirstat freezing
- Darla why are you sleeping
- Forza 5 release date 2021
- Is clustertruck split screen
- Girls of the golden west adams
- David winter tecdoc
- Video card driver standard vga graphics adapter download
But then we go over here to carbon 14 an isotope of carbon 12 protons are the same 6 protons and then we have 8 neutrons and 6+8 is 14 so our ratio neutron to proton ratio is 1.33 which is a bit high for the atomic numbers of such low magnitude 6 so what we're going to have to do is get rid of some of those neutrons so inside the nucleus what's going to happen is one of the neutrons is going to decompose and it's going to decompose into a proton and an electron. We like a ratio of 1 to 1 at low atomic numbers and carbon is low atomic number so it has a ratio of 1 to 1 so it's not going to actually undergo any sort of nuclear radiation that's totally okay it's safe. Well let's look at the stable atom we know that ca- carbon 12 has a 12 maximum number of 12 and 6 protons so neutrons is 6 and the protons is 6 making a total mass number of 12 and we have ratio is 1 to 1 which is what we like. Okay so beta decay is when it's above the band of stability so what happens then? Well the neutron proton ratio is too high it's above the band of stability meaning that there are too many neutrons okay so we have to do something about those neutrons inside that atom. Okay so why does this happen? Well the neutron and proton ratio is above the band of stability and if I go with band of stability over here it's this is a black kind of grayish line we'll make sure our neutron to proton ratio falls within that grayish line if it doesn't then something is going to happen it's either going to it's going to start radiating some sort of energy and things are going happen with the nuc- the nucleus to make it more stable and to lie within the band of stability.

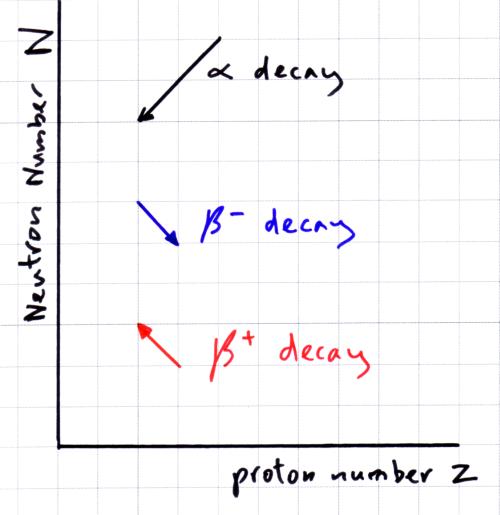
Alright when you're dealing with nuclear reactions or reactions that within the nucleus of an atom, there is several different types reactions that occur can occur one of them being beta decay and this is when a neutron decomposes into a proton and an electron.
